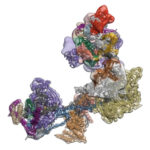
Eva Nogales, faculty scientist in Molecular Biophysics & Integrated Bioimaging (MBIB) Division and UC Berkeley professor of molecular and cell biology, led a team that captured freeze-frames of the changing shape of a huge macromolecular complex as it locks onto DNA and loads the machinery for reading the genetic code. The molecule, called transcription factor IID (TFIID), is critical to transcribing genes into messenger RNA that will later be used as blueprints to make proteins.
Photosynthesis, Like a Moss
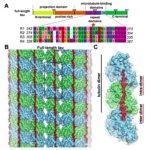
Using cryo-electron microscopy (cryo-EM), which allows an unprecedented level of resolution, Biosciences researchers compared the structure of photosystem I in the moss Physcomitrella patens with its structure in the small flowering land plant Arabidopsis thaliana, and in the green alga Chlamydomonas reinhardtii. Because moss evolved after algae but before vascular land plants, such comparisons can shed light on how plants evolved to move from the ocean to land.
Eva Nogales Honored by ASCB
The American Society for Cell Biology (ASCB) has selected Eva Nogales, a senior faculty scientist in Molecular Biophysics and Integrated Bioimaging (MBIB) and a Howard Hughes Medical Institute (HHMI) investigator and professor at UC Berkeley, as this year’s recipient of the Sandra K. Masur Senior Leadership Award. The award recognizes an outstanding scientist with a record of active leadership in mentoring both women and men in scientific careers. Nogales will be honored at the ASCB Annual Meeting in December 2018. Nogales was also elected by ASCB members to serve on the society’s executive committee as president-elect in 2019, president in 2020, and past president in 2021.
Tau-tally Microtubular: A Structural Model of Tau-Microtubule Interaction
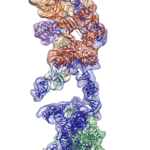
Researchers at Berkeley Lab and UC Berkeley have combined cutting-edge cryo-electron microscopy (cryo-EM) with computational molecular modeling to produce a near atomic-resolution model of the interaction between microtubules—crucial components of eukaryotic cell ultrastructure—and microtubule-associated proteins called tau. The model provides insight into how tau stabilizes microtubules, and what makes it dissociate to form tau aggregates, or “tangles,” in some neurological diseases—including Alzheimer’s disease—generally referred to as tauopathies.
Cryo-EM Reveals Structure of Human Telomerase
Regulation of the enzyme telomerase has been implicated in cancer, other human diseases, and aging, but progress towards therapeutic manipulation of telomerase has been hampered by the lack of high-resolution structural data. Now, researchers in senior faculty scientist Eva Nogales’ lab in Molecular Biophysics and Integrated Bioimaging (MBIB), in collaboration with UC Berkeley professor of Biochemistry, Biophysics and Structural Biology Kathleen Collins, have published a paper in Nature describing the 3-D molecular structure of the human telomerase enzyme.
- « Previous Page
- 1
- 2
- 3
- 4
- 5
- …
- 7
- Next Page »
Was this page useful?