Biophysicist Eva Nogales, a senior faculty scientist in the Molecular Biophysics and Integrated Bioimaging Division, is a 2023 recipient of the Shaw Prize in Life Science and Medicine. Nogales, who is also a UC Berkeley distinguished professor of biochemistry, biophysics, and structural biology, shares the prize with Patrick Cramer of the Max Planck Institute for Multidisciplinary Sciences in Germany. The pair were honored for pioneering structural biology techniques that enabled visualization of the protein machines responsible for gene transcription at the level of individual atoms.
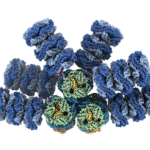
Cryo-EM Reveals Blueprint for Microbial Photosynthesis
Advances in cryogenic electron microscopy have enabled an international team of experts to visualize the structure of a cyanobacterial phycobilisome with nearly atomic resolution. The work, a collaboration among researchers at Michigan State University, UC Berkeley, Berkeley Lab, and the University of South Bohemia in the Czech Republic, was published in Nature. Knowing the position of different proteins and pigments helps scientist better understand this natural process and can inspire future applications in areas such as renewable energy and environmental remediation.
Two from Biosciences Area Named AAAS Fellows
The American Association for the Advancement of Science (AAAS) has announced that 564 of its members—among them four scientists at Berkeley Lab—have been elected Fellows as part of the 2021 class. The two newly named Fellows from the Biosciences Area are: Eva Nogales, a senior faculty scientist in the Molecular Biophysics and Integrated Bioimaging (MBIB) Division, and Scott Baker an affiliate with the Joint BioEnergy Institute (JBEI).
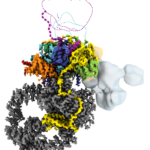
Structure of Human SAGA Protein Complex Solved
A team led by Eva Nogales, senior faculty scientist in the Molecular Biophysics and Integrated Bioimaging (MBIB) Division, has produced the first detailed 3D structure of human SAGA, a 20-piece molecular machine that’s crucial to life. The structure, reported in Nature Structural & Molecular Biology, revealed some unexpected differences between the human and yeast versions of SAGA and could guide the development of drugs to treat diseases that arise when this complex malfunctions.
Nogales Appointed to Royal Academy of Spain
Eva Nogales, a senior faculty scientist in the Molecular Biophysics and Integrated Bioimaging (MBIB) Division, was appointed as a foreign member attached to the Natural Sciences Section of the Royal Academy of Exact, Physical, and Natural Sciences of Spain. The Royal Academy, founded in 1847, is tasked with promoting study and research in the mathematical, physical, chemical, geological, and biological sciences, as well as disseminating the knowledge gained thereby. It is made up of a maximum of 72 permanent members, 144 corresponding members, and supernumerary members and foreign members. Nogales, who is also a Howard Hughes Medical Institute (HHMI) investigator and professor at UC Berkeley, obtained her bachelor’s degree in physics from the Universidad Autonoma de Madrid in Spain. Her research specialty involves using electron microscopy (EM) and image analysis, as well as biochemical and biophysical assays to gain mechanistic insights into crucial molecular processes in the life of eukaryotic cells.
- 1
- 2
- 3
- …
- 7
- Next Page »
Was this page useful?