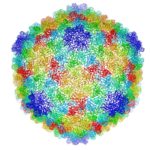
Cryo-electron microscopy is a critical tool used to advance biochemical knowledge. Now Pavel Afonine, research scientist, and Molecular Biophysics and Integrated Bioimaging Division Director Paul Adams have extended cryo-EM’s impact further by developing a new computational algorithm that was instrumental in constructing a 3-D atomic-scale model of bacteriophage P22 for the first time. Read more in the Berkeley Lab News Center.
The Strings That Bind Us: Cytofilaments Connect Cell Nucleus to Extracellular Microenvironment
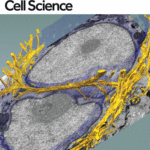
New images of structural fibers inside a cell appear in a study featured on the cover of the Journal of Cell Science special issue on 3D Cell Biology, published this month. The images, obtained by scientists in the Biosciences Area, show thread-like cytofilaments reaching into and traversing a human breast cell’s chromatin-packed nucleus.  It provides the first visual evidence of a physical link by which genes can receive mechanical cues from its microenvironment.
It provides the first visual evidence of a physical link by which genes can receive mechanical cues from its microenvironment.
The work leading up to the images began in the early 1980s when Biological Systems & Engineering’s Mina Bissell proposed the idea that gene expression and cell fate were dependent on their physical surroundings called extracellular matrix. The images were captured by Manfred Auer, staff scientist, and Ke Xu, faculty scientist, both in the Molecular Biophysics & Integrated Bioimaging Division. Read more at the Berkeley Lab News Center.
Workshop Focuses in on Electron Microscopy
An all-day workshop highlighting the Berkeley Lab’s capabilities in electron microscopy was held on Tuesday, October 11. Organized by Paul Adams (Biosciences Area), Peter Denes (Advanced Light Source) and Andy Minor (National Center for Electron Microscopy), the workshop highlighted recent advances in imaging a broad range of materials and biological samples at atomic, or near-atomic scales. In addition, it made evident the many opportunities that could come from integrating capabilities across the Laboratory. Paul Adams, Director of the Molecular Biophysics & Integrated Bioimaging Division, noted that the recent revolution in electron microscopy for biosciences has opened up many new avenues of research and exciting synergies with non-biosciences programs at the Lab. Read more in Glenn Roberts’ Science Short on the Berkeley Lab News Center.
New Technologies Fuel Cryo-EM’s Renaissance
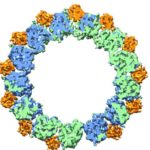
In a pair of breakthrough Nature papers published recently, researchers in Eva Nogales’ Lab at UC Berkeley and Berkeley Lab (Molecular Biophysics & Integrated Bioimaging Division) mapped two important protein functions in unprecedented detail: the role of TFIID, effectively improving our understanding of how our molecular machinery identifies the right DNA to copy; and how proteins unzip double-stranded DNA, which gives us insights into the first-key steps in gene activation.
These papers are representative of the renaissance currently under way in the cryo-electron microscopy (cryo-EM) field—driven primarily by the rise of cutting-edge electron detector cameras, sophisticated image processing software and access to NERSC supercomputing resources. Read the full story, written by Linda Vu of NERSC.
Scientists Image First Steps in DNA Transcription
Working at temperatures near absolute zero, scientists at Berkeley Lab and UC Berkeley have captured images of proteins at work in the early steps of DNA transcription. Eva Nogales, a senior faculty scientist at in the Molecular Biophysics & Integrated Bioimaging Division, UC Berkeley professor of biochemistry and a Howard Hughes Medical Institute (HHMI) investigator, led the new study, published today in the journal Nature.
This latest research comes seven weeks after Nogales unveiled an earlier milestone using cryo-EM technology to image details of transcription factor TFIID in gene expression. Read the full UC Berkeley press release on the newest Nature paper.
- « Previous Page
- 1
- …
- 4
- 5
- 6
- 7
- Next Page »
Was this page useful?