In two new studies, a team of researchers led by Eva Nogales, senior faculty scientist in the Molecular Biophysics and Integrated Bioimaging (MBIB) Division, has gained insight into the structure and function of molecules that act at the genetic level to give rise to different types of cells.
Detailed View of Immune Proteins Could Lead to New Pathogen-Defense Strategies
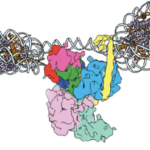
Berkeley Lab and UC Berkeley biologists used cryo-electron microscopy (cryo-EM) to resolve the structure of a protein complex, called an inflammasome, that is assembled within cells in response to incursion by a pathogen and functions as a beacon to summon immune system support. Inflammasome assembly is initiated when a protein called NAIP5 latches onto a flagellin molecule—a piece of the whiplike tail used by bacteria to propel themselves. Then several copies of another protein, NLRC4, join in to form the ring-shaped protein cluster. The researchers, led by Eva Nogales in Biosciences’ Molecular Biophysics and Integrated Bioimaging (MBIB) Division and Russell Vance in the Department of Molecular & Cell Biology at UCB, found that flagellin is in contact with six different parts of NAIP5. They further demonstrated that the multiple contact sites help prevent bacteria with minor mutations from eluding detection. The study was published in the journal Science. Read more in the Berkeley Lab News Center.
CRISPR Target Recognition Mechanism Illuminated Using Data From ALS
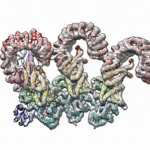
Using diffraction data obtained at Berkeley Lab’s Advanced Light Source (ALS) and at the Stanford Synchrotron Radiation Lightsource (SSRL), researchers in Biosciences’ Molecular Biophysics & Integrated Bioimaging (MBIB) division and at UC Berkeley have discovered how CRISPR-associated (Cas) proteins are able to recognize their target locations with such great specificity. X-ray crystallography was used to solve the structures of Cas1 and Cas2—responsible for DNA-snippet capture and integration—as they were bound to synthesized DNA strands designed to mimic different stages of the process. The resulting structures show how the system works in its native context as part of a bacterial immune system, and also inform the development of the CRISPR-Cas system as a general-purpose molecular recording device. Jun-Jie Liu, a joint postdoc in the labs of Jennifer Doudna and Eva Nogales, and Addison Wright, a graduate student in Doudna’s lab, were co-first authors on the paper, published in the journal Science. Read more in this ALS Science Highlight.
Biosciences Researchers Helped Lay Foundation for Nobel-Winning Cryo-EM
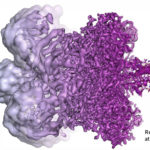
Several Berkeley Lab researchers contributed to the foundational work leading to the development of cryo-electron microscopy (cryo-EM), for which Jacques Dubochet, Joachim Frank, and Richard Henderson were awarded the 2017 Nobel Prize in Chemistry. Among those cited in the Scientific Background are Biosciences’ Bob Glaeser, Ken Downing, and Eva Nogales.
A Near-Atomic Resolution Map of a Key DNA Protein Complex
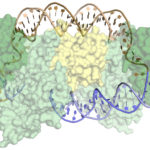
Molecular Biophysics and Integrated Bioimaging (MBIB) Division scientists led by Eva Nogales have resolved the 3-D structure of a critical human cellular protein complex involved in DNA transcription and repair at an unprecedented level of resolution. The complex, called transcription factor IIH (TFIIH), unzips the DNA double helix so that genes can be accessed and read. Malfunctions of the complex are associated with premature aging, cancer propensity, and a variety of other defects. One challenge with solving the structure of TFIIH has been that it exists in such minute amounts that it is difficult to produce and purify in large quantities. Moreover, once obtained, it may not form crystals suitable for X-ray diffraction. The researchers used cryo-electron microscopy (cryo-EM), a technique in which purified samples are flash-frozen at ultra cold temperatures, and which works even on very small quantities. “The fact that we resolved this protein structure from human cells makes this even more relevant to disease research,” said Nogales. Basil Greber, a postdoctoral fellow in Nogales’s lab, was first author on the study published in the journal Nature. Computational research scientist Pavel Afonine and MBIB Division Director Paul Adams also contributed to the project. Read more from the Berkeley Lab News Center.
- « Previous Page
- 1
- …
- 3
- 4
- 5
- 6
- 7
- Next Page »
Was this page useful?