Molecular Biophysics and Integrated Bioimaging (MBIB) Division scientists led by Eva Nogales have resolved the 3-D structure of a critical human cellular protein complex involved in DNA transcription and repair at an unprecedented level of resolution. The complex, called transcription factor IIH (TFIIH), unzips the DNA double helix so that genes can be accessed and read. Malfunctions of the complex are associated with premature aging, cancer propensity, and a variety of other defects. One challenge with solving the structure of TFIIH has been that it exists in such minute amounts that it is difficult to produce and purify in large quantities. Moreover, once obtained, it may not form crystals suitable for X-ray diffraction. The researchers used cryo-electron microscopy (cryo-EM), a technique in which purified samples are flash-frozen at ultra cold temperatures, and which works even on very small quantities. “The fact that we resolved this protein structure from human cells makes this even more relevant to disease research,” said Nogales. Basil Greber, a postdoctoral fellow in Nogales’s lab, was first author on the study published in the journal Nature. Computational research scientist Pavel Afonine and MBIB Division Director Paul Adams also contributed to the project. Read more from the Berkeley Lab News Center.
NIH Awards $9.3M for Further Development of PHENIX Structural Biology Software
The National Institutes of Health (NIH) has awarded $9.3 million to Berkeley Lab to support ongoing development of PHENIX, a software suite for solving three-dimensional macromolecular structures. Officially launched in 2000, the project is a collaboration among researchers based at Berkeley Lab, Los Alamos National Laboratory, Cambridge University, and Duke University. “The impetus behind PHENIX is a desire to make the computational aspects of crystallography more automated, reducing human error and speeding solutions,” said PHENIX principal investigator Paul Adams, director of Berkeley Lab’s Molecular Biophysics and Integrated Bioimaging Division (MBIB). Read more from the Berkeley Lab News Center.
Could This Enzyme Help Turn Biofuel Waste into Something Useful?
 Researchers at the Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) and Sandia National Laboratories working at the Joint BioEnergy Institute (JBEI) have resolved the protein structure of the enzyme LigM, which is utilized by the soil bacterium Sphingomonas to metabolize aryl compounds derived from lignin, the stiff, organic material that gives plants their structure. Their work is reported in this week’s Proceedings of the National Academy of Sciences. Read more in the Berkeley Lab News Center.
Researchers at the Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) and Sandia National Laboratories working at the Joint BioEnergy Institute (JBEI) have resolved the protein structure of the enzyme LigM, which is utilized by the soil bacterium Sphingomonas to metabolize aryl compounds derived from lignin, the stiff, organic material that gives plants their structure. Their work is reported in this week’s Proceedings of the National Academy of Sciences. Read more in the Berkeley Lab News Center.
Cryo-Electron Microscopy Achieves Unprecedented Resolution Using New Computational Methods
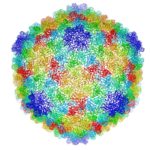
Cryo-electron microscopy is a critical tool used to advance biochemical knowledge. Now Pavel Afonine, research scientist, and Molecular Biophysics and Integrated Bioimaging Division Director Paul Adams have extended cryo-EM’s impact further by developing a new computational algorithm that was instrumental in constructing a 3-D atomic-scale model of bacteriophage P22 for the first time. Read more in the Berkeley Lab News Center.
Finding Diamonds in the Rough
New crystallography finding by JBEI and GLBRC benefits bioenergy industry
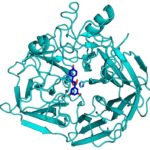 During the kraft process used to convert wood into wood pulp, the structural material lignin is partially converted into molecules like stilbene. Stilbenes are also naturally occurring in plants and some bacteria, and may play a role in plant pathogen resistance.
During the kraft process used to convert wood into wood pulp, the structural material lignin is partially converted into molecules like stilbene. Stilbenes are also naturally occurring in plants and some bacteria, and may play a role in plant pathogen resistance.
Currently, the deconstruction of plant biomass into cellulose and lignin is an expensive process. Lignin accounts for about 30 percent of plant cell wall carbon, and its conversion into chemicals or fuels could have a significant positive impact on the economics of processing lignocellulosic biomass. Enzymes capable of producing useful compounds from the breakdown of stilbenes and similar molecules could be employed for this. Collaborators from two of the Department of Energy Bioenergy Research Centers now have gained first-hand insight into how a stilbene cleaving oxygenase (SCO) carries out this unusual chemical reaction.
- « Previous Page
- 1
- …
- 5
- 6
- 7
- 8
- 9
- Next Page »
Was this page useful?