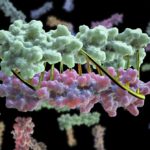
Bioscientists at the Advanced Light Source (ALS) at Berkeley Lab lent their expertise to a project led by scientists at the University of Washington to design proteins in the lab that zip together like DNA. The technique could enable the design of protein nanomachines to help diagnose and treat disease, allow for more precise engineering of cells, and perform a variety of other tasks.
Newly Characterized Toxin Gives Bacterium a Leg Up on the Competition
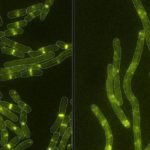
Bacteria deploy various biological mechanisms to fend off their competition, which are often other bacteria. In some cases, they secrete toxins in their fight for survival. A newly discovered toxin stands out from others in the battle for microbial domination. Marc Allaire, Molecular Biophysics & Integrated Bioimaging (MBIB) Division researcher, worked with a team led by Joseph Mougous of the Howard Hughes Medical Institute and University of Washington School of Medicine, to characterize this new toxin.
Recent Highlights from the Berkeley Center for Structural Biology Beamlines
Data collected at the Berkeley Center for Structural Biology (BCSB) in the Advanced Light Source (ALS) has provided new structural insights into an antibody that protects against the bacterium that causes meningitis and sepis; a protein that unwinds quadruple DNA/RNA helixes; and an antibody targeting interleukin-2 that may provide a means of treating autoimmune disorders.
Purdue Students Tour ALS
Corie Ralston, head of the Berkeley Center for Structural Biology and deputy director of the Molecular Biophysics and Integrated Bioimaging (MBIB) Division, and Mike Martin, Photon Science Operations Group Lead at the Advanced Light Source (ALS), led a tour of the ALS for a group of visiting biomedical engineering graduate students from Purdue University. Tammy Siemers, professional development and external relations specialist at Purdue University, arranges yearly ‘career immersion’ tours of companies and other places where graduate students might end up working one day. “The reason I chose ALS (and Lawrence Berkeley National Laboratory in general) is that I knew they were doing research in areas that our students would be interested in,“ said Siemers. “We had a great visit to ALS.”
The tours–which have taken place in Washington, DC, Boston, and Seattle previously–are important for the students to meet scientists and learn more about their options after graduate school. “I also like for students to gain networking skills and to learn about valuable resources and potential collaborations on this tour,” Siemers added. “ALS definitely has some valuable resources for research that we weren’t even aware of prior to this trip.”
Open and Shut: Pain Signals in Nerve Cells
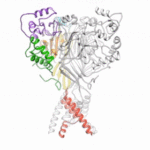
Using protein crystallography at the Advanced Light Source (ALS), investigators at Oregon Health and Science University visualized an acid-sensing ion channel, ASIC1a, in it’s elusive closed state for the first time. An important cell membrane protein in the central nervous system, ASIC1a plays a role in sensing pain and in forming memories of fear. The researchers used protein crystals of ASIC1a channels purified in the closed state and compiled X-ray data from ALS Beamline 5.0.2 and the Advanced Photon Source at Argonne National Lab to build a model of the closed channel structure. Defining the closed structure enabled the researchers to generate a comprehensive molecular model of how ASIC1a toggles between the closed, open, and desensitized states.
Read more from the ALS.
- « Previous Page
- 1
- …
- 4
- 5
- 6
- 7
- 8
- 9
- Next Page »
Was this page useful?