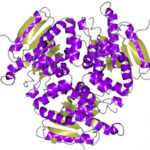
The Advanced Light Source hit a structural biology milestone in May 2018 with the help of their eight structural biology beamlines. Users of these beamlines have now collectively deposited over 7000 proteins into the Protein Data Bank (PDB), a worldwide, open-access repository of protein structures. The 7000th ALS protein structure (PBD accession number 6C7C) is an enzyme from Mycobacterium ulcerans (strain Agy99), solved with data from Beamline 5.0.2 in the Berkeley Center for Structural Biology. The enzyme is of interest to the researchers from the Seattle Structural Genomics Center for Infectious Disease (SSGID), whose mission is to obtain crystal structures of potential drug targets on the priority pathogen list of the National Institute of Allergy and Infectious Diseases (NIAID). Beamline 5.0.2, the first protein crystallography beamline at the ALS, came online in 1997. Read more in the ALS Feature.
ALS Enables Structural Determination of Respiratory Virus-Antibody Complex
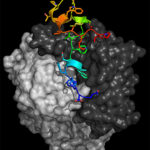
Using diffraction data collected at cryogenic temperature at the Advanced Light Source (ALS) Beamline 8.3.1, University of California Santa Cruz (UCSC) scientists, led by assistant professor of biomolecular engineering Rebecca DuBois, determined the three-dimensional atomic structure of a major surface protein of the respiratory syncytial virus (RSV): RSV G. Medical researchers have been trying to develop a vaccine for RSV, which causes serious respiratory disease in infants and older adults, for more than 50 years without success. But by leveraging the work of collaborators at Trellis Bioscience, who isolated protective human antibodies targeting RSV G, an attachment protein that allows the virus to stick to lung cells, the UCSC scientists were able to show that these protective antibodies target a section of the protein called the central conserved domain that is the same in all strains of the virus. The findings, published March 9 in Science Immunology, point to a promising route for designing a vaccine effective against a broad range of RSV strains. Read more from the UC Santa Cruz News Center.
Modified Antibody Clarifies Tumor-Killing Mechanisms
At the Advanced Light Source (ALS), researchers studied an antibody that was modified to activate a specific pathway of the immune system, demonstrating its value in killing tumor cells. The work provides a platform for disentangling the effects of different immune-system pathways and could lead to the design of improved cancer immunotherapies. The protein crystallography data in the study, which showed how an extreme twist in the modified antibody structure led to its selectivity, was collected at ALS Beamline 5.0.3 , which is part of the Biosciences’ Berkeley Center for Structural Biology; and at the Advanced Photon Source. Go to the ALS website to learn more.
‘Minimalist’ Machine Learning Algorithms Analyze Images from Sparse Data
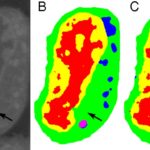
Typical machine learning methods used to analyze experimental imaging data rely on tens or hundreds of thousands of training images. But Daniël Pelt and James Sethian of Berkeley Lab’s Center for Advanced Mathematics for Energy Research Applications (CAMERA) have developed what they call a “Mixed-Scale Dense Convolution Neural Network” (MS-D) that “learns” much more quickly from a remarkably small training set. One promising application of MS-D is in understanding the internal structure and morphology of biological cells to identify, for example, differences between healthy and diseased cells. In one such project in Carolyn Larabell’s lab, the method needed data from just seven cells to determine the cell structure.
Crystallization Screen Created by Berkeley Lab Biosciences Scientists Reaches the Market
 X-ray crystallography has been the most successful technique used to solve macromolecular structures, contributing several thousand new entries to the Protein Data Bank (PDB) every year. The protein crystal is the critical starting point for X-ray data collection, and consequently, its properties are correlated with the quality of the data and the level of detail that can be extracted for a macromolecular structure. However, proteins require solutions of specific composition to form crystals for structure determination studies. These specifications are usually determined from exposing the protein to several different solutions in a crystallization screen.
X-ray crystallography has been the most successful technique used to solve macromolecular structures, contributing several thousand new entries to the Protein Data Bank (PDB) every year. The protein crystal is the critical starting point for X-ray data collection, and consequently, its properties are correlated with the quality of the data and the level of detail that can be extracted for a macromolecular structure. However, proteins require solutions of specific composition to form crystals for structure determination studies. These specifications are usually determined from exposing the protein to several different solutions in a crystallization screen.
A team of researchers in the Molecular Biophysics and Integrated Bioimaging (MBIB) Division led by Paul Adams and Jose Henrique Pereira have developed a new crystal screen, the Berkeley Screen, with 96 conditions proven to be highly effective at producing crystals for structural determination. The Berkeley Screen is now available to the wider crystallography community commercially.
- « Previous Page
- 1
- …
- 13
- 14
- 15
- 16
- 17
- 18
- Next Page »
Was this page useful?