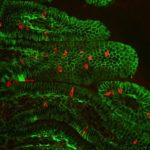
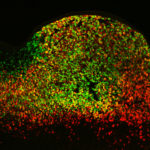
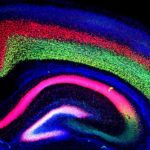
Berkeley Lab geneticist Len Pennacchio and his team helped a group of Israeli clinical researchers solve the mystery of a rare inherited disease that causes extreme, sometimes fatal, chronic diarrhea in children. The nearly decade-long investigation not only led to the discovery of a novel protein-coding gene that is critical for intestinal function, but also expanded our understanding of regulatory sequences in the human genome. The results were recently published in Nature.
Gene Enhancers Are Important Despite Apparent Redundancy
Though once thought to be “junk DNA,” enhancers are extremely important, regulating the expression of specific genes that ultimately determine a cell’s properties and functions. However, there are many more enhancers than genes and their relationship is unclear. A team of scientists in the Environmental Genomics & Systems Biology Division have turned their attention to this relationship and the overall importance of enhancers to development. By better linking the genomic complement of an organism with its expressed characteristics, their work offers new insights that further the growing field of systems biology, which seeks to gain a predictive understanding of living systems.
Researchers Collaborate to Create, Characterize Mouse Model of Autism
Using a genetic mouse model developed at Berkeley Lab with CRISPR/Cas9 editing technology, University of California, Davis, researchers led by Alexander Nord have examined the developmental impact of a specific mutation found in some rare cases of autism spectrum disorder (ASD). The project began while Nord—now an assistant professor with the Center for Neuroscience at UC Davis—was a postdoc in the Mammalian Functional Genomics Laboratory of Berkeley Lab co-authors Axel Visel, Len Pennacchio, and Diane Dickel. In a paper published June 26 in the journal Nature Neuroscience, the researchers report that mice with a loss-of-function mutation in one copy of the CHD8 gene have increased brain volume and cognitive impairment, similar to that seen in people with the same mutation. CHD8 encodes a protein responsible for packaging DNA in cells, which in turn controls gene expression during development. Read more from UC Davis.
Finding Our Way Around DNA
Biosciences researchers collaborated with a team at the Salk Institute that developed a computational algorithm that integrates two different data types to make locating key regions within the genome more precise and accurate than other tools. The team’s method, recently described in Proceedings of the National Academy of Sciences, could help researchers conduct vastly more targeted searches for disease-causing genetic variants in the human genome, such as ones that promote cancer or cause metabolic disorders. Joseph Ecker, a Howard Hughes Medical Institute investigator and director of Salk’s Genomic Analysis Laboratory, was senior author of the study. Diane Dickel, Axel Visel and Len Pennacchio of the Environmental Genomics & Systems Biology Division, were co-authors, along with other researchers at the Salk Institute, UC San Diego, and UC San Francisco. Read more about the study in the Salk Institute press release.
Was this page useful?