Most people know that smoking causes cancer, and many are aware of the dangers of secondhand smoke. Unlike the smoke you can see or smell in the moment, thirdhand smoke (THS) refers to the toxic residues that tobacco smoke leaves behind on furniture, walls, clothing, carpets, and dust—often long after the last cigarette has been extinguished. Two recent studies, both involving scientists from the Biological Systems and Engineering (BSE) Division at Berkeley Lab, shed new light on the potential dangers of this invisible contaminant, not just for cancer risk, but for brain health as well. These findings provide compelling evidence that environmental exposures interact with genetic variation to shape health outcomes.
A Double Threat: Initiating and Promoting Tumors
The first study, a comprehensive review published in the journal Environment & Health, took a systematic look at THS through the lens of classical cancer biology. Led by corresponding authors Bo Hang, formerly a staff scientist, now a retiree affiliate, and Jian-Hua Mao, a senior scientist, both in BSE, in collaboration with colleagues in the UC San Francisco Department of Medicine, the paper asks a fundamental question: does THS help start the cancer process, help it spread, or both?
Cancer typically develops in stages. Tumor “initiation” refers to the moment when a cell’s DNA is permanently damaged, setting it on a path toward malignancy. “Promotion” happens later, when those damaged cells are encouraged to multiply and avoid the body’s normal defenses. The team presents compelling evidence from the scientific literature of THS operating at both stages.
Earlier work from the Berkeley Lab team was the first to demonstrate that THS causes DNA strand breaks in human cells, a finding that helped shape California and New York legislation targeting THS in home-based daycare centers. THS contains a toxic cocktail of more than 100 identified chemicals, including tobacco-specific nitrosamines—compounds that form when nicotine reacts with indoor air pollutants—as well as polycyclic aromatic hydrocarbons and volatile organic compounds. These substances can directly damage DNA, forming structural abnormalities that, if unrepaired, can trigger the mutations needed to initiate tumor development.
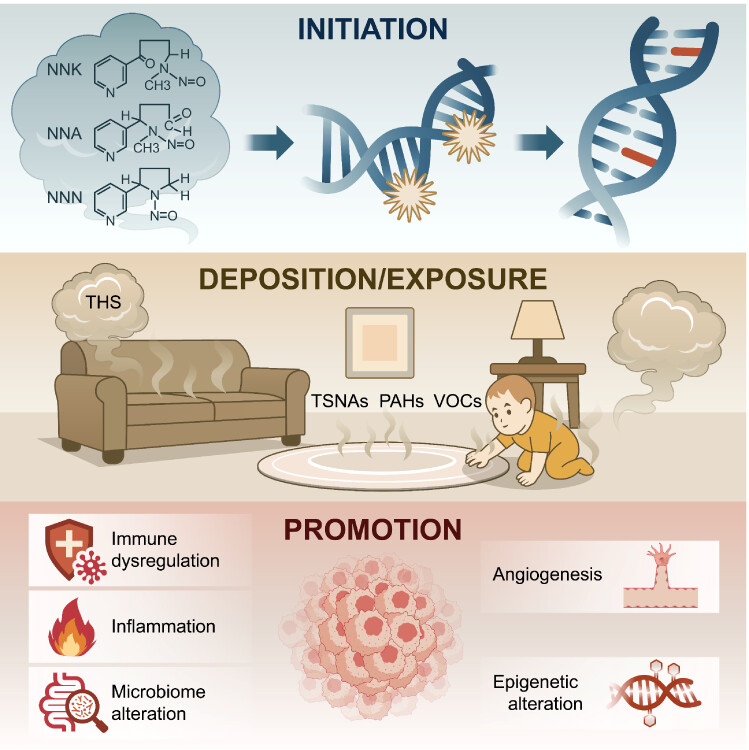
Beyond direct DNA damage, THS also appears to work as a tumor promoter through more subtle mechanisms. Chronic exposure can trigger persistent low-grade inflammation, generate reactive molecules that destabilize cellular machinery, and alter gene expression through modifications that affect how genes are turned on or off. Together, these effects create an environment in which genetically damaged cells are more likely to survive and grow. The team notes that while the DNA damage associated with initiation is irreversible, THS’s promotional effects may be partially reversible if exposure stops, highlighting the potential value of environmental remediation efforts.
Nature and Nurture: Sensitivity Shaped by Genetics
A companion study published in Environment International expands the picture of THS’s harms well beyond cancer. Led by BSE staff scientist Hang Chang and research scientist Jamie Inman as senior authors, the experiment interrogated the extent to which genetic makeup shapes vulnerability to THS’s affects the brain—specifically anxiety and memory.
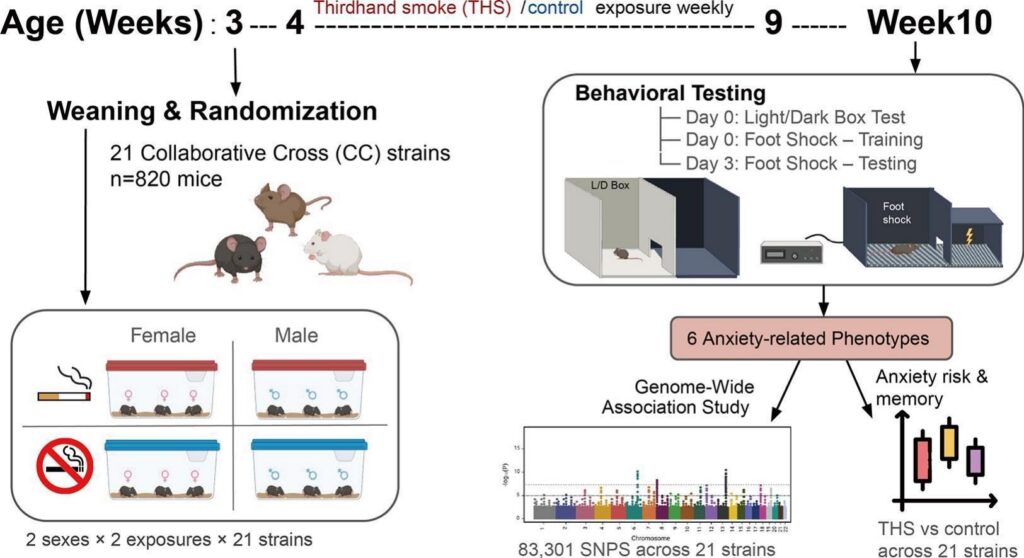
The team used a sophisticated rodent resource called the Collaborative Cross (CC), a diverse population of mouse strains that collectively captures roughly 90% of the genetic variation found in laboratory mice, making it a powerful stand-in for studying human diversity. A total of 820 mice from 21 genetically distinct strains were exposed to THS-contaminated bedding during early life, then assessed using standardized behavioral tests for anxiety and memory.
THS did not affect all mice the same way. In female mice, some strains showed increased anxiety after exposure, while others showed decreased anxiety. And in males, no significant effects on anxiety were detected. Memory impairments were similarly uneven: some strains showed deficits in both sexes, while others showed effects only in one sex.
To dig deeper, the researchers performed a scan of hundreds of thousands of genetic variants and identified thousands of locations in the genome associated with anxiety and memory. Dozens of these locations showed a meaningful interaction with THS exposure, pointing toward genes involved in how neurons form connections, how synapses maintain plasticity, and how signals travel between brain cells.
Understanding the complex interactions between genes and environment presents a major scientific challenge requiring integration of large and diverse datasets spanning genomics, environmental measurements, behavioral phenotypes, and molecular biology, noted Chang. “AI-driven frameworks provide the ability to integrate these diverse layers of information, revealing previously hidden relationships between environmental exposures and biological systems,” he said.
Using machine learning, the team built predictive models that could classify individual animals by anxiety and memory risk with reasonable accuracy, demonstrating that a genetic fingerprint of THS sensitivity may be achievable. “By elucidating how THS residues interact with genetic susceptibility and brain function, this study underscores the growing role of AI-enabled, data-driven science in addressing some of the most pressing environmental health challenges of our time,” Chang added.




