Researchers in the Biological Systems and Engineering (BSE) Division recently published two studies that will help oncologists more precisely understand the state of their patients’ disease or their risk for cancer relapse. As with many diseases, cancer can be challenging to predict and in some cases, impossible to treat. This work, however, is pushing the boundaries of how science and artificial intelligence (AI) can be used to better understand the risks and outcomes of cancer in human health.
While the two studies were conducted by separate groups and focused on different types of cancer, BSE researchers Hang Chang and Jian-Hua Mao led the studies in both publications. Chang, a research scientist, and Mao, a senior scientist, largely focus their work on developing and validating machine learning techniques in cancer research.
“AI techniques introduce the potential for precision treatment; they can help doctors provide more accurate, robust, and cost-effective patient diagnosis and treatment,” Chang said.
The first study, published in June in Neuro-Oncology, highlighted a machine-learning tool that can identify cellular morphometric biomarkers (CMBs), or cells that forecast cancerous behavior, from tissue sample images. These biomarkers have the potential to provide value far beyond the clinical setting, Chang said, and may even reveal new avenues to pursue for therapeutic development.
This work focused on patients with a type of malignant brain tumor known as lower-grade gliomas (LGG) that originate in support cells called glia. Researchers accessed nearly 1,000 images of LGG tissue samples from the publicly available Cancer Genome Atlas.
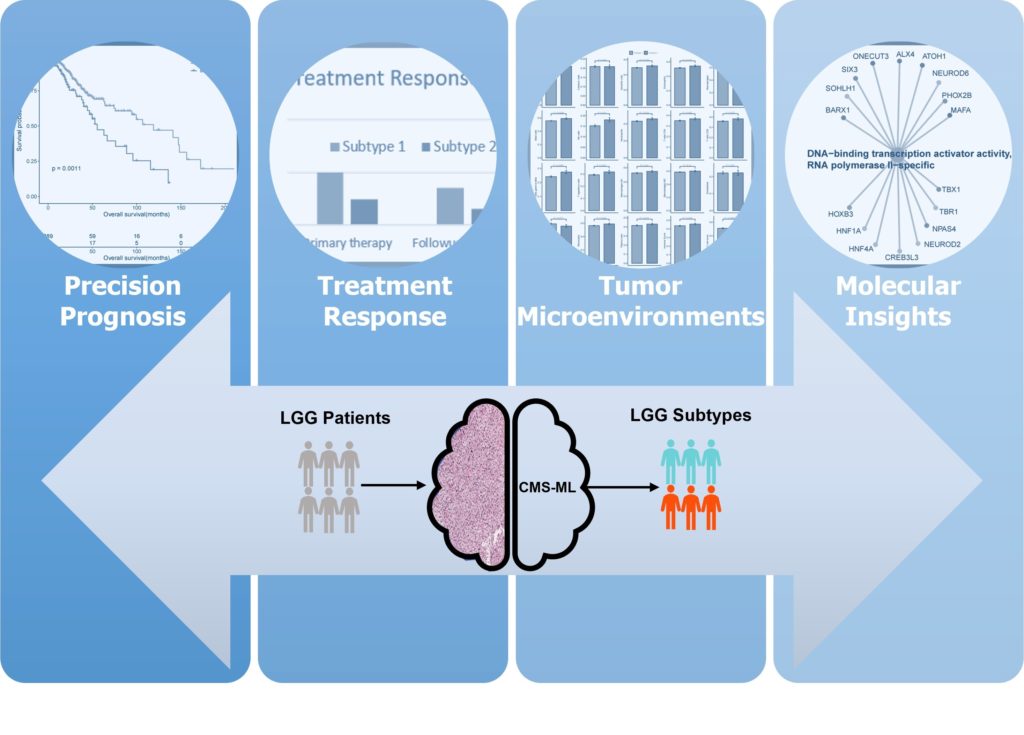
With their collaborators, the team built and tested a series of data-processing steps that resulted in a pipeline that can automatically identify CMBs from images of tissue samples. The group then defined and validated LGG subtypes based on CMBs associated with specific molecular alterations, immune micro-environment, prognosis, and treatment response.
The second study, published in July in World Journal of Clinical Oncology, built upon the CMB pipeline to include additional data points to assess a patient’s risk of breast cancer recurrence. This multimodal machine learning approach also incorporated tumor microbiome and genetic data into AI models to more precisely predict a patient’s breast cancer risk.
The Biosciences Area researchers and those from the University of Salamanca in Spain used whole histological slide images from the Cancer Genome Atlas to determine whether a patient is at a low, intermediate, or high risk for breast cancer recurrence. With these three types of data integrated into a machine-learning pipeline, the group developed a scoring system called iCEMIGE, or integration of cell morphometrics, microbiome, and gene biomarker signatures.
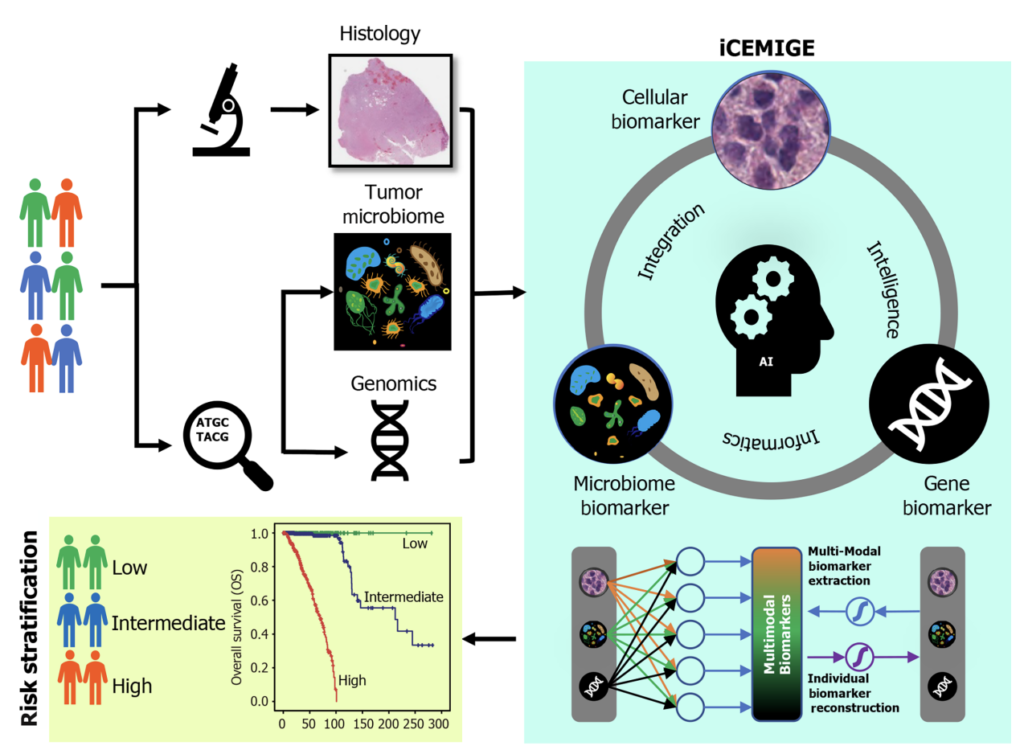
This work demonstrated that, in the case of AI and breast cancer prognosis, the more information brought into the equation, the stronger the predictive outcome. iCEMIGE was able to significantly improve risk prediction of breast cancer patients as compared to the prognosis based solely on one type of data. The researchers are now looking to further analyze and validate these results with the goal of applying these AI pipelines to other types of cancer as well.
Collectively, these findings introduce new tools that can provide a deeper understanding of the cellular changes associated with two types of cancer. With a better understanding of risk, clinical oncologists can help patients pinpoint which treatment options may work best for them.
Other BSE researchers involved in this work include: Xiaoqing Jin, Xiao-Ping Liu, Xuan-Yu Mao, Antoine M. Snijders, and Xu Yang.
This work was supported by the National Cancer Institute of the National Institutes of Health and the Department of Defense.




