Scientists have for the first time reengineered a building block of a geometric nanocompartment that occurs naturally in bacteria. The new design provides an entirely new functionality that greatly expands the potential for these compartments to serve as custom-made chemical factories. The work was led by Cheryl Kerfeld, who holds joint appointments with Berkeley Lab’s 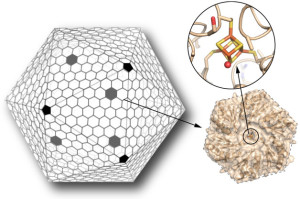 Molecular Biophysics and Integrated Bioimaging Division, UC Berkeley and the MSU-DOE Plant Research Laboratory at Michigan State University. Markus Sutter, a senior research associate in Kerfeld’s group at Berkeley Lab, collected the X-ray diffraction data used in this study in the Berkeley Center for Structural Biology at the Advanced Light Source. Read more at the Berkeley Lab News Center.
Molecular Biophysics and Integrated Bioimaging Division, UC Berkeley and the MSU-DOE Plant Research Laboratory at Michigan State University. Markus Sutter, a senior research associate in Kerfeld’s group at Berkeley Lab, collected the X-ray diffraction data used in this study in the Berkeley Center for Structural Biology at the Advanced Light Source. Read more at the Berkeley Lab News Center.
Biosciences Area Scientists Participate in World Economic Forum
Faculty biochemist Jennifer Doudna and Biosciences Area Associate Laboratory Director Jay Keasling attended the World Economic Forum in Davos, Switzerland, last week, to bring their unique perspectives to world leaders. Both researchers were named to Al Aribya’s “45 top scientists at Davos you should know about” list.
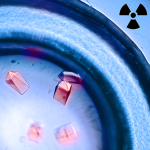
A New Pathway for Radionuclide Uptake
Synthetic radionuclides, such as the transuranic actinides plutonium, americium, and curium, present severe health threats as contaminants, and understanding the scope of the biochemical interactions involved in actinide uptake into cells is instrumental in managing human contamination. Recently, scientists have reported that an iron-binding protein called siderocalin can also bind and transport actinides into cells, a major advance in understanding the biological chemistry of radioactive metals. The research, which included X-ray crystallographic studies in the Berkeley Center for Structural Biology (Beamlines 5.0.1 and 5.0.2) at the Advanced Light Source (ALS), opens up new avenues of research into strategies for remedial action in the event of possible human exposure to nuclear contaminants. In addition, siderocalin, the protein studied, was selected as “Molecule of the Month” by the Protein Data Bank’s educational portal. Read more in the ALS Science Highlight.
Got It Down Cold: Cryo-Electron Microscopy Named Method of the Year
In his blog late last week, NIH Director Francis S. Collins highlighted Cryo-EM Microscopy, the Method of the Year 2015. Collins gave a retrospective analysis of how far the method has come over the past decade, and credits most of the advances to improvements in the technology available, particularly computational methods for image construction and direct electron detectors that can fix the blurring problem caused by molecules moving around in the electron beam. With these and other changes, the number of structures determined using this method have increased substantially in just the past year. Molecular Biophysics & Integrated Bioimaging’s Robert Glaeser got a shout-out for his Nature Methods commentary, which says that owing to the laws of physics, we can expect more good things.
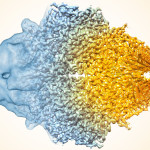
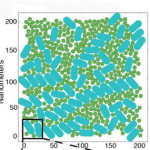
Seeing the Big Picture in Photosynthetic Light Harvesting
Graham Fleming, chemist senior faculty scientist in Molecular Biophysics & Integrated Bioimaging, led the creation of the first computational model that simulates the light-harvesting activity of the thousands of antenna proteins that would be interacting in the chloroplast of an actual leaf. The results from this model point the way to improving the yields of food and fuel crops, and developing artificial photosynthesis technologies. Read more at Berkeley Lab News Center.
- « Previous Page
- 1
- …
- 69
- 70
- 71
- 72
- 73
- …
- 75
- Next Page »
Was this page useful?