Using the SLAC National Accelerator Laboratory’s Linac Coherent Light Source (LCLS) X-ray laser, an international collaboration led by scientists at Berkeley Lab and SLAC captured the all four stable oxidation states of photosystem II— plus two transitional states—at natural temperature and the highest resolution to date.
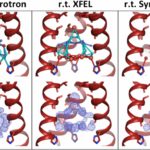
Room Temperature XFEL Provides Clearest View Yet of Water Networks in Influenza M2 Proton Channel
Molecular Biophysics & Integrated Bioimaging (MBIB) Division scientists Aaron Brewster, Nicholas Sauter, and James Fraser were part of an international team led by William DeGrado at UCSF that used an X-ray free-electron laser (XFEL) source to visualize the arrangement of water molecules inside the influenza matrix 2 (M2) channel at room temperature. The M2 channel of influenza A is essential for the reproduction of the flu virus, making it a target for therapeutics, and it is also a model system for studying how protons are transported across a membrane bilayer. The XFEL method overcomes the limitations of previous crystallographic structures obtained using synchrotron radiation with cryocooling. While cryocooling helps to preserve crystals against rapid radiation damage, it imparts an artificially higher degree of order of the water molecules than structures obtained near room temperature. By using room temperature XFEL to study the M2 channel at various pH conditions, the researchers have gained a more accurate picture of the behavior of water molecules and their role in proton transport in these channels. The study was published in the Proceedings of the National Academy of Sciences.
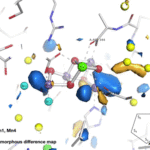
X-rays Capture Unprecedented Images of Photosynthesis in Action
An international team of scientists is getting closer to discovering how plants split water during photosynthesis and produce nearly all of the oxygen in our atmosphere. Thanks to unprecedented, atomic-scale images of a protein complex found in plants, algae, and cyanobacteria captured by ultrafast X-ray lasers, researchers conducted atomic-level experiments to help delineate the mechanism of this system that also yields the protons and electrons used to reduce carbon dioxide to carbohydrates later in the photosynthesis cycle. The effort to uncover the secrets of this protein complex, photosystem II, was led by Vittal Yachandra and Junko Yano in the Molecular Biophysics & Integrated Bioimaging (MBIB) Division and the team’s findings were published this week in Nature.
X-rays Reveal New Path In Battle Against Mosquito-borne Illness
MBIB researchers were part of a team that used SLAC’s X-ray free-electron laser (XFEL) – the Linac Coherent Light Source (LCLS), a DOE Office of Science User Facility – to get atomic views of the toxin BinAB, used as a larvicide to control mosquito-borne diseases such as malaria, West Nile virus and viral encephalitis. The structure of this bacterial toxin was solved using de novo phasing: the protein crystals were tagged with heavy metal markers, tens of thousands of diffraction patterns were collected using the XFEL, and the information was combined to obtain a three-dimensional map of the electron density of the protein. The Berkeley team, headed by Senior Scientist Nicholas Sauter, acquired and processed data for the study, published in Nature last week.
Instrumentation Advances Expand the Reach of X-ray Free Electron Lasers
Femtosecond crystallography (FX) is especially suitable for studying radiation sensitive enzymes that require metals for their function, as the extremely short and bright X-ray pulses can produce a diffraction image before any atomic motions can occur in the crystal. This cutting edge method is capable of extending our capacity to study smaller, more fragile crystals and determine the catalytic structures of biologically relevant macromolecules.
Was this page useful?